Glass Ionomer sealants (GI) explained: composition, chemical adhesion, fluoride release/recharge, moisture tolerance, and antimicrobial effects. Evidence on clinical effectiveness, ideal indications (partially erupted molars, high caries risk, field programs), RMGI/HV-GI advances, step-by-step placement, maintenance, limitations, and best practices aligned with ISO/FDA-ready workflows by XDENT LAB
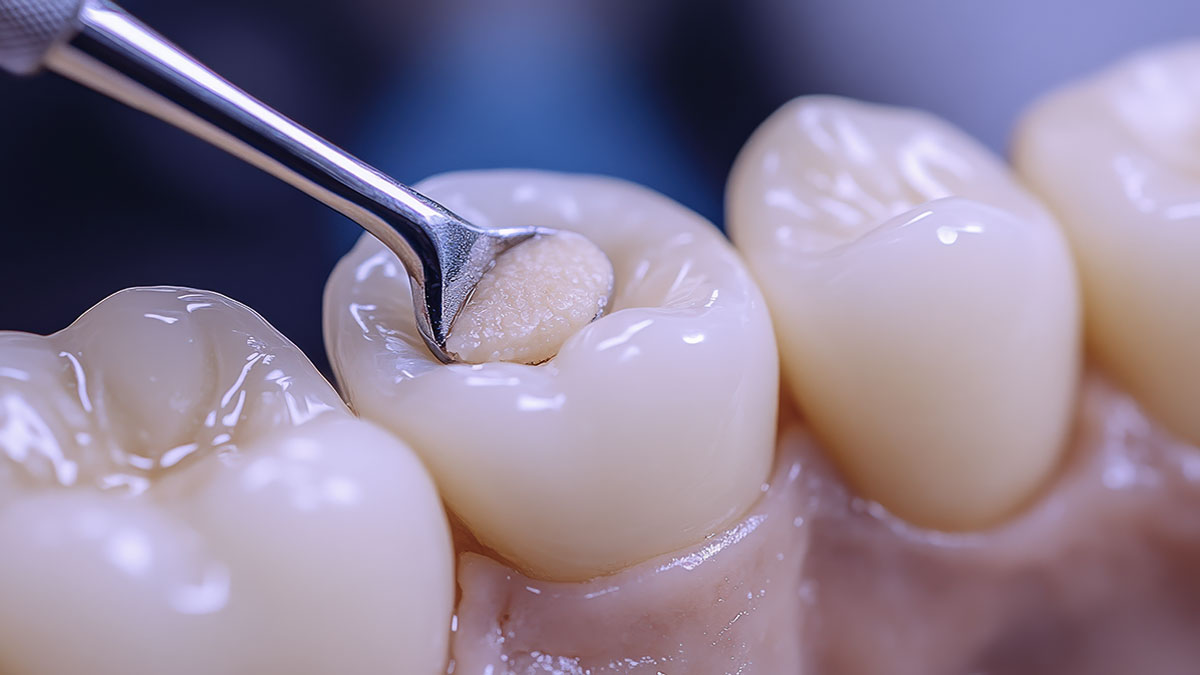
Table of contents [Show]
- Overview
- Composition & Setting Mechanism
- Key Properties & Clinical Significance
- Clinical Effectiveness & Indications
- GI vs. Resin-Based Sealants: Practical Comparison
- Formulation Advances
- Cost-Effectiveness & Public Health Impact
- Clinical Protocol: Best Practices
- Limitations & How to Mitigate
- XDENT LAB Perspective (FDA/ISO Aligned)
- Key Takeaways
Overview
Glass Ionomer (GI) sealants are a cornerstone of preventive dentistry when isolation is difficult and fluoride support is desirable. Unlike resin-based sealants that rely on micromechanical retention, GI forms a chemical bond to tooth structure, releases and recharges fluoride, and tolerates moisture - Making it highly effective for partially erupted molars, young patients, and community programs. Below is a comprehensive analysis of composition, properties, clinical performance, and best practices, aligned with XDENT LAB’s quality and compliance stance.
Composition & Setting Mechanism
GI sealants are water-based, ion-releasing cements that set via an acid–base reaction.
Core Chemistry
Powder: Fluoroaluminosilicate glass (Ca/Sr/Ba aluminosilicate with fluoride).
Liquid: Aqueous polyacrylic acid (or related polyalkenoic acids).
Additives: Set modifiers, handling agents, radiopacifiers.
Setting Phases
Initial: Acid attacks glass → releases Ca2+, Al3+, F–.
Gelation: Metal ions cross-link polyacrylic acid chains.
Maturation: Continued cross-linking and water binding.
Adhesion: Chemical ionic bonding to hydroxyapatite (enamel/dentin).
What this means in practice: reliable bonding without a separate adhesive, and a fluoride reservoir built into the sealant.
Key Properties & Clinical Significance
GI’s properties map directly to high-yield preventive outcomes in challenging scenarios.
Chemical Adhesion
Bonds to enamel and dentin via carboxylate–calcium interactions.
Reduces dependence on perfect etch-and-dry fields compared with resins.
Fluoride Release & Recharge
Initial burst (24–48 hours), followed by sustained low-level release.
Rechargeable from toothpaste/varnish → ongoing cariostatic effect.
Even partially retained GI can continue providing localized fluoride benefit.
Moisture Tolerance
Hydrophilic system functions in moist environments.
Clinical edge in partially erupted molars, uncooperative children, field dentistry.
Biocompatibility & Antimicrobial Effects
Low cytotoxicity and pulpal irritation.
Unfavorable environment for bacteria during set; fluoride-mediated biofilm inhibition.
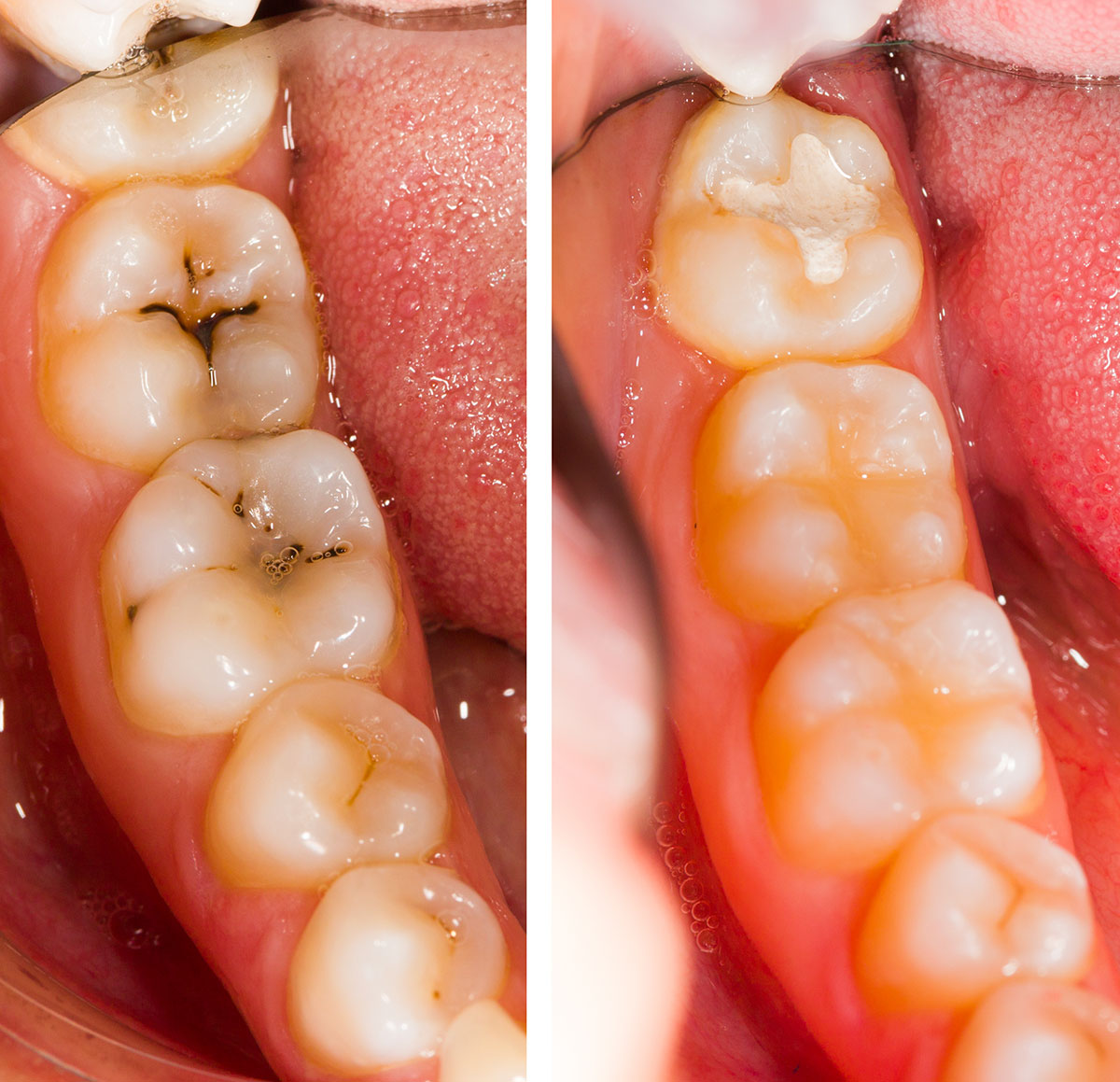
Clinical Effectiveness & Indications
GI sealants are validated for caries prevention even when retention isn’t perfect.
Effectiveness Highlights
Caries risk reduction on occlusal surfaces is substantial.
Long-term studies: similar caries prevention to resin-based sealants despite lower retention.
Survival analyses: no significant difference in caries incidence versus resin in many cohorts.
Ideal Use Cases
Partially erupted permanent molars with difficult isolation.
Young or behaviorally challenging patients.
High caries risk patients requiring fluoride support.
School- or community-based programs and resource-limited settings.
Interim protection until ideal resin placement conditions are possible.
GI vs. Resin-Based Sealants: Practical Comparison
| Feature | Glass Ionomer (GI) | Resin-Based Sealants |
|---|---|---|
| Adhesion | Chemical bond to tooth | Micromechanical (etch/bond) |
| Moisture control | Tolerant | Requires dry field |
| Fluoride | Release + recharge | Limited (unless fluoride-containing) |
| Retention | Traditionally lower, improving with HV/RMGI | Higher short-term retention |
| Wear resistance | Lower than resin | Higher |
| Technique sensitivity | Lower | Higher |
| Best fit | Partially erupted, field, high caries risk | Fully erupted, excellent isolation |
Key takeaway: choose GI when isolation is compromised or fluoride benefit is strategic; choose resin for maximum retention under ideal isolation.
Formulation Advances
Modern GI variants mitigate historical drawbacks while preserving strengths.
Resin-Modified Glass Ionomer (RMGI)
Dual set: acid–base plus light-cure.
Improved early strength, faster set, better retention than conventional GI.
Retains fluoride release and chemical adhesion.
High-Viscosity (HV) GI
Smaller particles, higher powder–liquid ratio, modified acids.
Enhanced wear resistance, faster set, better retention than earlier GI generations.
Strong fit for ART and field programs.
Cost-Effectiveness & Public Health Impact
Lower technique sensitivity reduces chairside failures and re-seal rates.
Minimal equipment requirements favor mobile and field delivery models.
In community programs, GI’s moisture tolerance and fluoride effect improve scalability and outcomes.
Clinical Protocol: Best Practices
Standardized steps enhance success and reproducibility across operators.
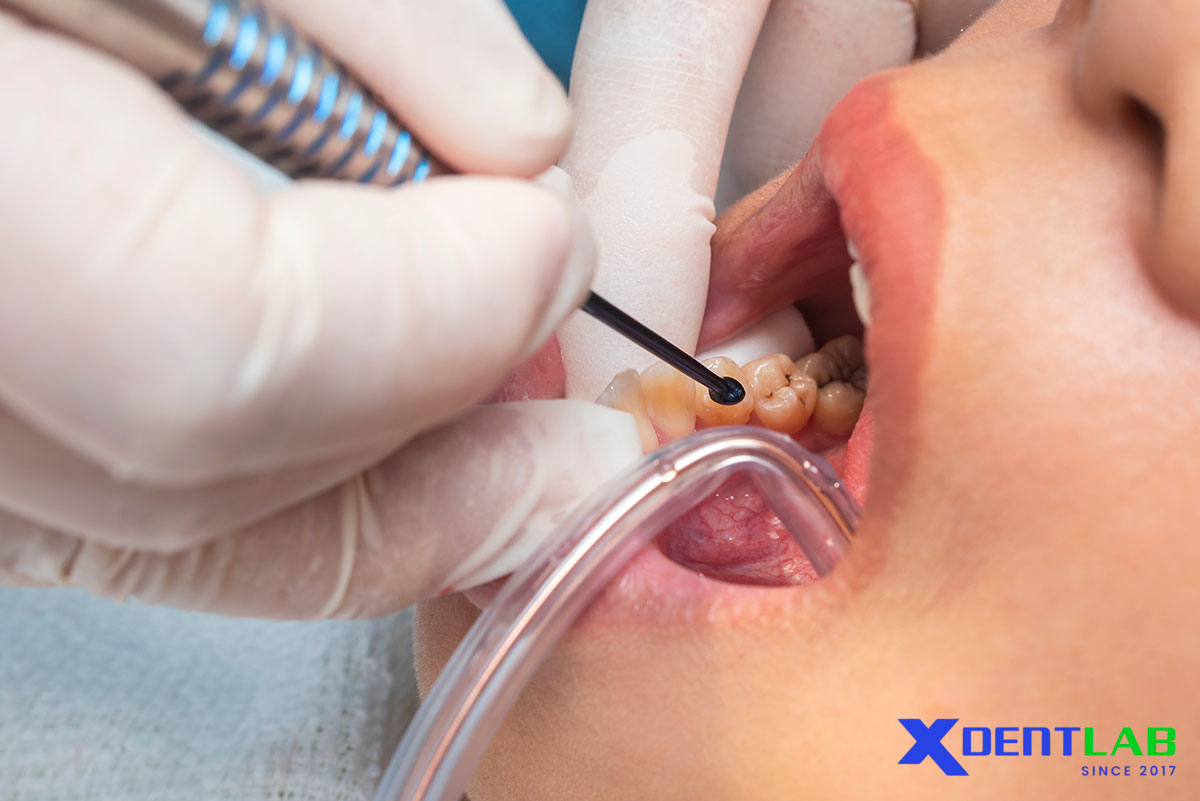
Indications
Partially erupted molars, limited cooperation, high caries risk, field settings, interim sealing.
Placement Technique
Clean fissures (prophy paste or dry brush).
Condition enamel with 10–20% polyacrylic acid for 10–20 seconds; rinse, gently dry (do not desiccate).
Mix per IFU; apply with pressure into pits and fissures.
Protect surface during initial set (petroleum jelly or resin coat per IFU).
Verify and adjust occlusion after full set and surface protection.
Provide fluoride hygiene guidance to support recharge.
Maintenance
Six-month recalls for retention and integrity.
Reapply if partial loss and caries risk persists.
Reinforce fluoride exposure (toothpaste, professional treatments).
Limitations & How to Mitigate
Lower wear and retention versus resin: consider RMGI or HV-GI, surface coatings, recall reinforcement.
Esthetics (opacity, staining): set expectations; consider resin where esthetics dominate.
Setting sensitivity to excess moisture or dehydration: protect surface during initial set.
XDENT LAB Perspective (FDA/ISO Aligned)
XDENT LAB integrates evidence-based material selection with audit-ready documentation for U.S.-market expectations.
Operational Playbook
Material curation: RMGI and HV-GI for moisture-challenged indications and ART-style deployments.
IFU adherence: standardized conditioning, set protection, and occlusal checks.
QA documentation: batch traceability, IFU compliance, operator SOPs, recall recommendations.
Training assets: chairside quick guides for pediatric and community settings.
Compliance posture: ISO-driven process control; FDA-aligned documentation for U.S. partners.
Outcome: predictable preventive performance in non-ideal conditions, with consistent records for quality audits and practice risk management.
Key Takeaways
GI sealants provide chemical adhesion, fluoride release and recharge, and moisture tolerance—translating to robust caries prevention when isolation is challenging.
Modern RMGI and high-viscosity formulations improve retention and wear while preserving GI’s preventive advantages.
In many scenarios, GI’s caries-preventive effect matches resin-based sealants despite lower retention; material choice should be indication-driven, not assumption-driven.
XDENT LAB’s lab-to-lab framework ensures standardized protocols, documentation, and compliance, supporting U.S.-standard preventive outcomes at scale.
XDENT LAB is an expert in Lab-to-Lab Full Service from Vietnam, with the signature services of Removable & Implant, meeting U.S. market standards – approved by FDA & ISO. Founded in 2017, XDENT LAB has grown from local root to global reach, scaling with 2 factories and over 100 employees.. Our state-of-the-art technology, certified technicians, and commitment to compliance make us the trusted choice for dental practices looking to ensure quality and consistency in their products.

Our commitments are:
100% FDA-Approved Materials.
Large-Scale Manufacturing, high volume, remake rate < 1%.
2~3 days in lab (*digital file).
Your cost savings 30%.
Uninterrupted Manufacturing 365 days a year.
Contact us today to establish a strategy to reduce operating costs.
--------❃--------
Vietnam Dental Laboratory - XDENT LAB
🏢 Factory 1: 95/6 Tran Van Kieu Street, Binh Phu Ward, Ho Chi Minh City, Vietnam
🏢 Factory 2: Kizuna 3 Industrial Park, Can Giuoc Commune, Tay Ninh Province, Vietnam
☎ Hotline: 0919 796 718 📰 Get detailed pricing
Share this post: