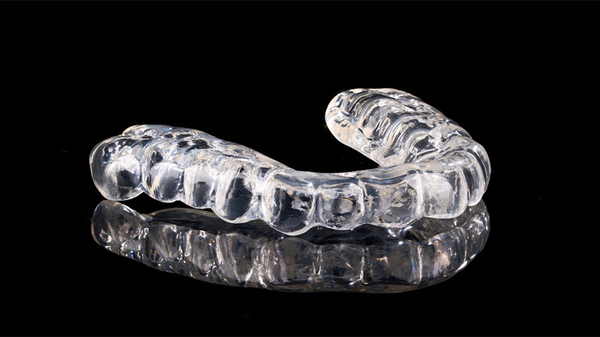
When it comes to protecting teeth from grinding or clenching, night guards play a vital role. Understanding the materials used in night guard production is essential for both dentists and patients, as...

Learn key polishing considerations for hard and soft night guards, including heat control, surface finish, fit accuracy, polishing compounds, and material-specific techniques.

An overview of income and labor costs for dental laboratory technicians across major global markets. How does the average income of dental lab technicians differ between the United States, Europe, Chi...

Explore how light-curing resin is being applied in 3D printed nightguards, from flexible materials and digital workflows to post-processing techniques that improve accuracy, durability, and overall pe...
Explore silicone polymers in dentistry, including their properties, applications, advantages, limitations, and role in impressions, prosthetics, and soft liners.

Explore 5 prominent dental laboratories in South Korea known for digital dentistry, CAD/CAM precision, implant prosthetics, esthetic restorations, and advanced lab workflows.

Learn how returned restoration cases increase waste in dental labs through material loss, opportunity cost, production bottlenecks, and damage to client trust.

Learn how vestibular depth in dentistry affects oral anatomy, denture retention, implant planning, periodontal health, clinical assessment, and treatment decisions.

Learn when to use a lingual bar in RPD, its design features, indications, limitations, and clinical considerations in mandibular major connector design.

Explore polyamide (nylon) in thermoplastic dentistry, including its properties, applications, advantages, limitations, and role in flexible dentures and removable prosthetics.

Learn mandibular connectors in RPD, including functions, types, design principles, and a practical step-by-step framework approach.

Vietnam dental lab outsourcing manufacturers and suppliers benefit from strategic location, open policies, tax advantages, and strong logistics. XDENT LAB delivers reliable outsourcing solutions for g...


